The lungs, as we know, are organs essentially not unlike large sacks filled with lots of smaller sacks. They facilitate the ingress of life giving oxygen to our biological systems. Each lobe of the lungs contains increasingly smaller chambers and pathways, the smallest being the alveoli. The lungs are central to the respiratory system and facilitate the exchange of outward bound waste gas from the body and inward bound gasses such as oxygen. This is called gas exchange, or pulmonary gas exchange and is the fundamental mechanism of breathing. Without it, life is impossible under normal circumstances. When pulmonary gas exchange is compromised by injury or diesease, life theatening complications rapidly ensue. To preserve life, oxygen transport must be maintained and this can be achieved, as will be demonstrated, by increasing ambient pressure and oxygen concentration, and by extension the partial presdure of the oxygen being breathed.
Overview
Beginning at the nose and mouth, the respiratory system conveys air inward towards the lungs. Air (gas) enters through the nose and mouth and then passes down the throat (pharynx), through the voice box (larynx), and onward toward the lungs. The entrance to the larynx is covered by a small flap of tissue called the epiglottis. This closes automatically when swallowing to prevent food and drink from entering the airways.
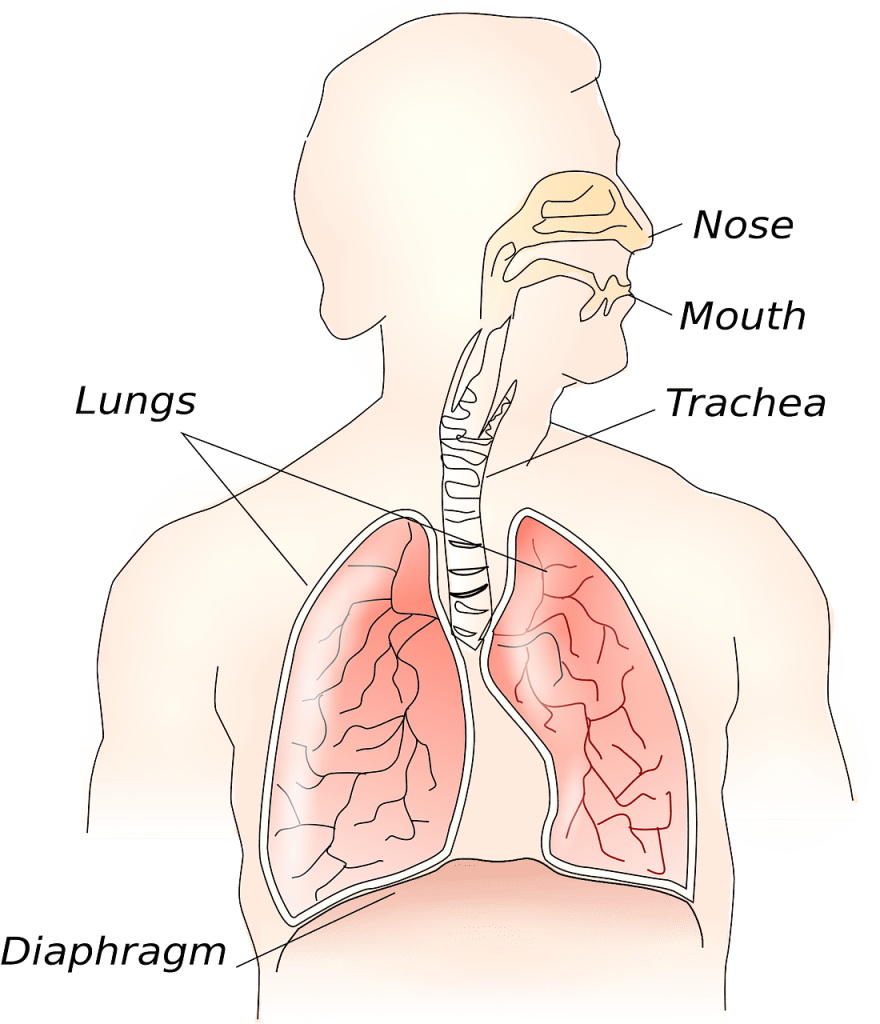
The windpipe (trachea) is the largest airway. The trachea branches into two smaller airways: the left and right bronchi, which lead to the two lungs.
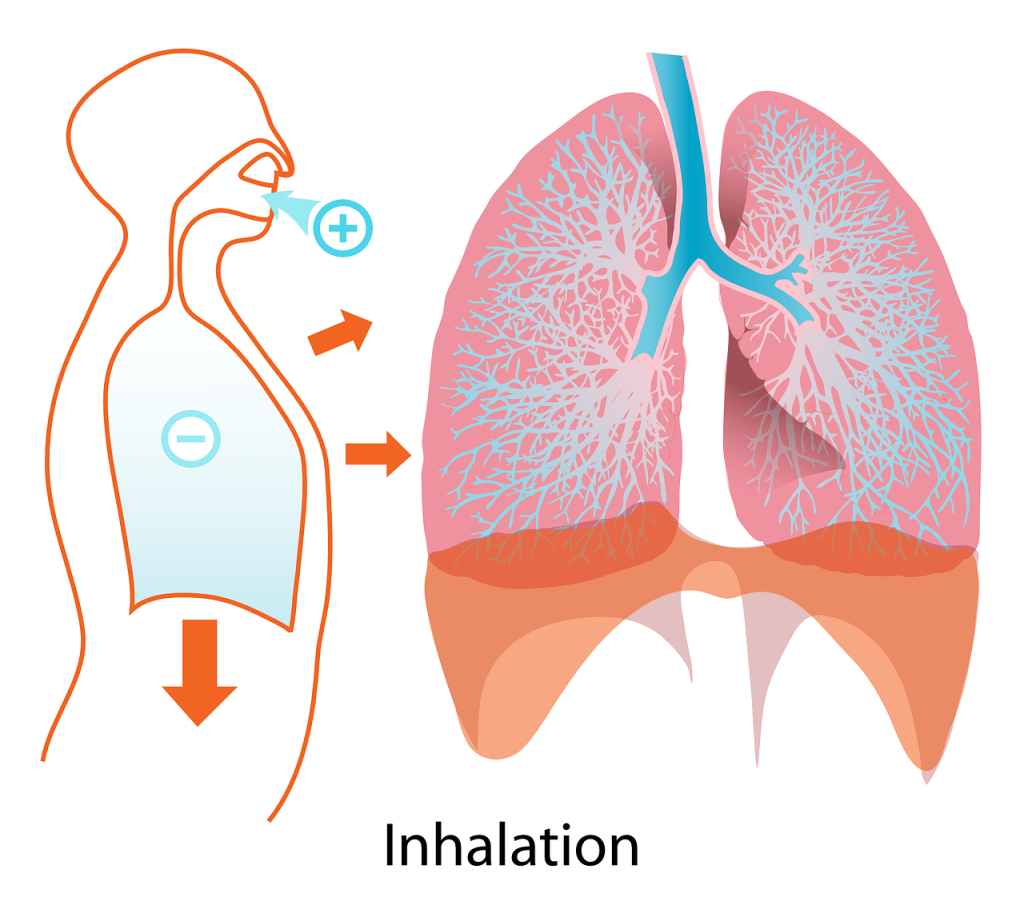
From there the trachea carries air inward as we breathe in to the right and left bronchus, and on to the bronchiole on both sides, eventually reaching the air sacs called the alveoli. All this is driven by the contracting of the diaphragm which creates an area of lower pressure in the lungs drawing air in. The reverse happens when we breathe out again, with the help of a flexing diaphragm now causing a higher pressure in the lungs carrying air outward. [29] Anatomy and Physiology – Rice University [30] US Navy Diving Manual 7th Edition 2016
Our focus for this discussion is what happens in the air sacs or alveoli. This is where air (gas) meets liquid. It is where our singular most important biological process and mechanism exist. The process which allows us to continue living and allows oxygen in and carbon dioxide out of the body. Without it, life would quickly cease. It is also where we absorb metabolically inert or non-reactive gases such as atmospheric nitrogen, argon or even helium, as in the case of very deep diving mixes. (Dives deeper than 50 meters are generally undertaken on a helium and oxygen mix called heliox, or a combination of air and helium called Trimix. Oxygen – Nitrogen – Helium).
On one side of the alveoli wall there is blood and on the other there is air/gas. Between them is tissue as little as one cell across, which is coated with surfactant on the air, (alveolar space) side, rendering it essentially ‘waterproof’. This surfactant prevents blood from passing into the lungs. It also facilitates the lowering of the surface tension at the point of the gas/liquid interface. Explained in the graphic below. This is important to remember as when reading the articles on oxygen tolerance and toxicity , and how it is greatly misunderstood in general, and has become probably the singularly most preventative factor in the willingness of professionals to administer hyperbaric and even normobaric oxygen therapy/treatment in patients who would benefit greatly from it.
“Pulmonary surfactant is a complex and highly surface-active material composed of lipids and proteins which is found in the fluid lining the alveolar surface of the lungs. Surfactant prevents alveolar collapse at low lung volume and preserves bronchiolar patency during normal and forced respiration (biophysical functions). In addition, it is involved in the protection of the lungs from injuries and infections caused by inhaled particles and micro-organisms”
Eur Respir J. 1999 Jun.
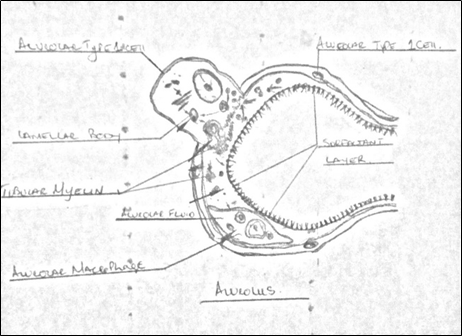
“Pulmonary surfactant is a mixture of lipids and proteins which is secreted by the epithelial type II cells into the alveolar space. Its main function is to reduce the surface tension at the air/liquid interface in the lung.”
Biochimica et Biophysica Acta 2000
It’s at this time we must become familiar with the new term describing gas pressure as previously mentioned. That is, “Gas tension”.
You will have previously read the words, “In the gas phase”. “Gas phase” simply means that while a gas is actually a free gas, it is in the gas phase. When we breathe in, a gas dissolves into the blood, and in the case of oxygen, dissolves into the blood and then attaches itself to the haemoglobin. It ceases to be in the gas phase at this pint and beocmes a dissolved gas.
Similarly, when carbon dioxide dissolves into water to make fizzy pop or soda water, it achieves a gas “tension” within a liquid as a dissolved gas.
Gas pressure no longer in the “gas phase” is referred to as gas tension, as it becomes solute in the solution in this example, blood or water respectively. Gas tension is the pressure a gas exerts in solution within a liquid. It is the equivalent of gas pressure in the gas phase.
In medical terms, “blood-gas”, or tissue gas tension, will almost always be measured in millimetres of mercury (mmHg). A unit of particular familiarity when considering the measurement of blood pressure using mercury calibrated manual sphygmomanometers. Conversely, in diving, we commonly use atmospheres to measure pressure either in whole atmospheres or fractions of atmospheres. They are simply different units of measurement, with mmHg being a far more accurate and precise unit used in medicine to determine very accurate measurements of pressure and tensions, as opposed to the comparatively brutish unit of atmospheres in diving.

When reading medical papers and studies, pressures or tensions of dissolved gas in tissues will almost always be presented in mmHg and this is referring to pressure that dissolved gas exerts in tissue and liquid, and by virtue of that, the environment (the actual ambient atmosphere). The conversion factor is 760 mmHg for 1 atmosphere (1atm or 1ata). It is this pressure that facilitates the movement and transport of gas in our biological systems.
Accordingly, barometric pressure at sea level, when normal, is 760 mmHg. For the purposes of measurements in diving and hyperbarics we use atmospheres absolute, bars, psi (pounds per square inch) and so on. Since this article won’t cover specific quantities (grams or molar mass) of gas but rather the theory of mechanisms, normal pressure reference units will be favoured. Quantities of dissolved gasses would be measured in grams, micrograms etcetera.
This gas tension can be accurately measured in tissues using a device capable of measuring oxygen saturation below the skin. This is known as transcutaneous oxygen measurement (transcutaneous oximetry) (TCOM or TcPO2). With the TcPO2 standing for: Transcutaneous Pressure of Oxygen. The term pressure, or partial pressure of oxygen, is used considerabley within my writing. It is a necessary concept and analysis method in HBOT because it allows for the understanding of, and the measurement of how much oxygen is saturated into the tissues, rather than only measuring the heamoglobin saturation. This concept and physical law is better described in the aricle PHYSICS, JOHN DALTON AND THE CUMBRIAN CONNECTION .
As mentioned before, tissue saturation trumps heamoglobin saturation since tissue saturation is the end goal of heamoglobin saturation. Without adequate tissue saturation, heamoglobin saturation is largely irrelevant. It’s worth noting that adequate tissue saturation can be achieved without heaoglobin as discussed in the article LIFE WITHOUT BLOOD.
Gas and liquid are both fluids and they interact according to a variety of factors including density and pressure. A dissolved gas in a liquid will still behave according to the gas laws previously explained in Henry’s Law and Daltons Law and Boyles Law. It will still exert its partial pressure in the liquid as it does in the gas phase. In the gas phase, reducing pressure will cause gas to simply expand, in the liquid phase it will cause it to come out of solution and return to gas form, it will “off-gas” in accordance with Henry’s Law. We simply refer to it as gas tension in the liquid, and gas pressure in the gas form.
The following explanation is the basis for decompression theory, and originates in the works of visionary, John Scott Haldane (1860 – 1936). A Scottish physiologist famous for intrepid self-experimentation, which led to many important discoveries about the human body and the nature of gases. He is sometimes referred to as the “Father of Oxygen Therapy”. [32] Encyclopaedia Britannica 2018 He developed some of the first decompression schedules or tables and his work in altitude studies in climbers, balloonists and the early stages of flight, were revolutionary to say the least. To divers he is the father of diving as well. We still dive today on the basis of the Haldanean model. This applies to hyper and normo baric conditions as well. In all of the sciences involved in aerospace, altitude and diving physics, respiration and gas exchange is central.
A wrist-mount dive computer is possible because of the work of JS Haldane. He is cited in almost every diving manual ever written. Although his work has been built upon, and some of the thinking on oxygen exposure has been challenged, the basic science is still Haldanean, and it all begins with gaseous interchange or gas exchange and the gas laws discussed above. Gas exchange itself is not specifically a diving science however diving science describes it best, at least to my sensibilities since it is all driven by gradients which is the underlying science in pulmonary gas exchange.
Pressure Gradients:
Dealing with oxygenation first, the mechanism responsible for the actual carriage of oxygen into the blood, the reason we breathe at all, is called an inward pressure gradient, responsible for sustaining oxidative cell metabolism and life itself, is explained as follows:
When we breathe air into our lungs and that air mixes with the last breath we breathed out, the fraction of oxygen (%) in that air increases since our last breath will have a reduced oxygen concentration having been partially used by the body, thus creating a differential in the tension (pressure) of the oxygen in the tissue (the blood), and the gas (the alveola air). This differential is referred to as a pressure gradient.
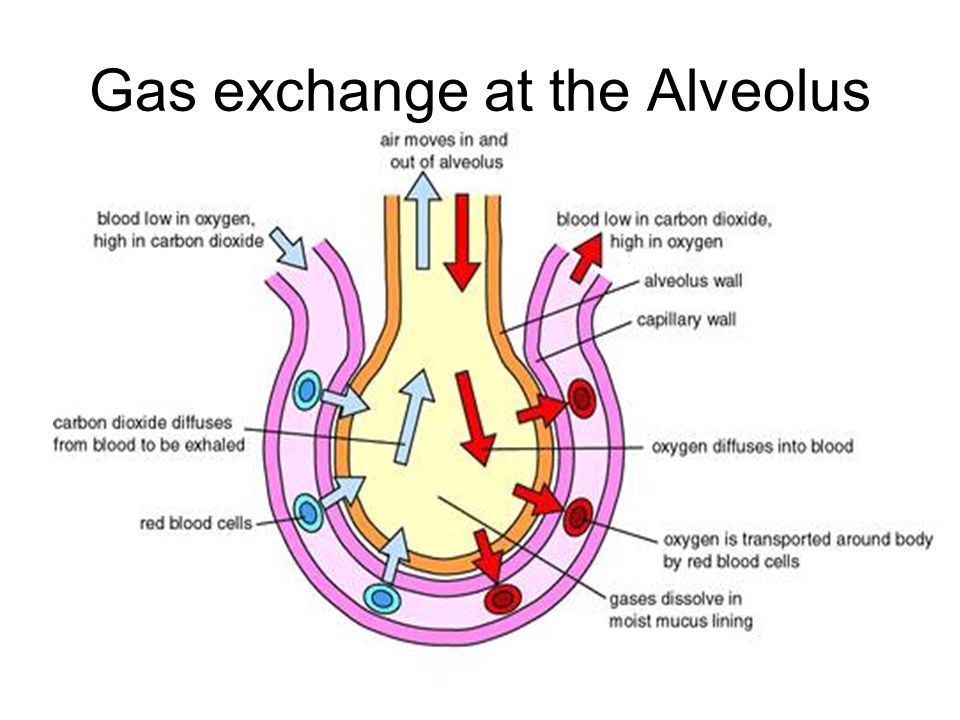
In the presence of a gradient that can be described as “inward”, or in other words, higher pressure on the air side than the blood side of the alveolar wall, Henry’s law applies and the oxygen will seek equilibrium and dissolve into the liquid until equilibrium is reached. It will “in-gas”. When equilibrium is achieved a maximum plasma load is met and no more oxygen will dissolve into the blood. Those oxygen molecules which just dissolved into the blood will then attach themselves to haemoglobin in the red blood cells (RBC’s) and will be carried through the circulatory system. [29] Anatomy and Physiology -The Process of Breathing Chapter 22 – Rice University [28] Pulmonary Gas Exchange 1998 [30] United States Navy Diving Manual 7Th Edition 2016
The oxygen then travels with the bloodstream until it comes to a tissue or cell which exhibits a lower gas tension that it itself does, and in the same way, dissolves into that tissue or cell plasm. It’s a downstream effect. This will continue until equilibrium is again achieved. As we know, in the case of oxygenation, equilibrium fluctuation is what drives the process. Oxygen tension and pressures will constantly switch back and forth, facilitating what is termed “oxygen transport”. Owing to the metabolic nature of a living organism, oxygen is constantly being metabolised which changes its concentrations and pressures, or tension. This also directly changes the carbon dioxide tension as well.
The only way to increase plasma load is to increase the saturation capacity of plasma by increasing the pressure at which a gas is breathed.
We exist at a point of near equilibrium in normal atmospheric pressure. We are essentially saturated to 1 atmosphere absolute in terms of gas saturation with minor fluctuations as we breathe in and out. Because oxygen is reactive (metabolically active), a portion of it is consumed by cells in the oxidative metabolic process and the gas tension in the cell drops compared to the passing blood. As the cells produce carbon dioxide (CO2) as a by-product of oxidative metabolism, the tension of the CO2 rises above that of the passing blood and a reverse gradient now exists for CO2. Known as an “Outward Gradient”, this will drive the removal of metabolic by-products like CO2 from the cells across the cell membrane into the blood stream. At the same time, the lower oxygen tension in the cell causes an inward gradient once more for oxygen which now has a higher tension in the blood stream. The CO2 will travel back to the lungs via the circulatory system and when it encounters the alveolar wall, it will have a higher gas tension than its counterpart in the alveolar air, once again Henry’s Law applies and it comes out of solution from the blood. In other words, it “out-gasses” and again exists in the gas phase in the lungs. The lungs then breath out this CO2 and the process continues.
Essentially there is never a point of absolute equilibrium. It’s a see-saw process that goes on for the duration of our lives. Quite beautiful in its complexity yet simple enough. With every breath we breathe, the saturation level changes back and forth as the gas tensions change back and forth, facilitating breathing, oxygen transport, waste elimination and life.
A word on metabolically inert gases is relevant at this point: Metabolically inert gases such as nitrogen (N2) and helium (He) are metabolically inert and as such, don’t react within the body. These gases will also seek equilibrium as we breathe, until such time as the gas tension in the blood and the gas pressure in the air is equal. We are then said to be saturated in as far as that gas goes for that given ambient pressure (normally whatever atmospheric pressure may be at our location.
As long as our surrounding or ambient pressure remains the same, that level of saturation will remain as it is indefinitely. In the main, and for those that remain largely at ambient barometric pressure, that’s where its ends. You needn’t know about it other than out of interest. It’s when ambient pressure changes, such as would happen in activities such as diving or flying that things change within the body and can lead to unfavourable effects. For example, diving accidents and illness, and altitude sickness. In the case of metabolically active oxygen, the contrary applies. The additional oxygen results in the upregulation of our natural physiological processes of healing and rebuilding that has been going on since we were born.
And thats how breathing works. Or medically put, pulmonary gas exchange. Its about the pressure. Its also the reason behind altitude sickness despite the air still containing normal oxygen percentages. More on that in the article ALTITUDE, THE HYPEROXIC HYPOXIA PARADOX, AND THE COMMON SENSE PARADOX.
PRESERVE OXYGEN TRANSPORT – PRESERVE LIFE
© Hayden Dunstan



Terrific article
LikeLike