
Following the first two installments of the toxicity discussion, we address the sometimes misunderstood and consequently overstated subject of pulmonary toxicity. This relates to the effect that long term oxygen breathing on a repetitive basis can have on the lungs.
Being a fairly long article, and not all readers want all of the science, I’ll begin with the takeaway. If the science proves too long winded then this is the short version.
Pulmonary oxygen toxicity refers to the effect breathing pure oxygen, or gases enriched with oxygen, can have on the lungs in cases of long term exposure. It is said to result from inflammation, and ultimately fibrosis in the lung tissue following extended periods of “UNINTERRUPTED” oxygen breathing. It is rarely encountered in diving owing to the fact that it is something that occurs over fairly long periods of time. Much longer periods than even those encountered in standard HBOT treatment protocols. Ordinarily dive times and the fairly long exposure limits prevent a diver or chamber occupant from coming anywhere near range of pulmonary toxicity or intolerance.
Notwithstanding that, there are specific treatment types such as saturation treatments and the saturation environment where long term exposure is a given, where exposure must remain with established tolerable limits.

This is explained further:
It may become a consideration as well in cases of extended decompression, or where chamber treatments or surface decompression (diving) procedures are adopted following a dive in which some exposure has already accrued. It should also be considered in those cases where a chamber occupant may be the subject of multiple exposures in a single day or over many days. In these cases the tracking of a measurement unit called the Oxygen Tolerance Unit (formerly “oxygen toxicity units” and abbreviated to “OTU’s”) becomes necessary.
Following extreme exposures, pulmonary toxicity can present as mild chest pain, wheezing, a dry cough and general respiratory discomfort and can eventually lead to tissue damage and fibrosis (scarring) in the lungs in extreme cases if not managed properly. This tissue damage in many cases is reversible, especially with research into drugs which facilitate protection on the increase, however in rare and severe cases it can cause permanent damage. (United States Navy Diving Manual 7th Edition 2016)
Pulmonary tolerance has historically been measured in “Oxygen Tolerance/Toxicity Units” or OTU’s. The word toxicity is again interchangeable with tolerance since it’s not so much a toxic response as an intolerance response when this does occur. It was also historically measured by the “UPTD”, the “unit pulmonary toxicity dose”. In cumulative measurements it is referred to as the “CPTD”, the “cumulative pulmonary toxicity dose”. Basically, all of these come back to the number of units an individual is exposed to and they refer to ways of tracking OTU’s, the unit measure of exposure. Exposure limits are based on the number of OTU’s a subject is exposed to.
One OTU is equal to breathing pure oxygen at sea level for one minute. (Tolerating Exposure to High Oxygen Levels – Repex and Other Methods Hamilton 1989_
And by that we do mean pure 100% oxygen. It is unlikely that a free flow mask on a ward or ambulance will deliver anywhere near that purity. Only a tight fitting oral nasal mask can do that. Accordingly, when you’re told you’re breathing pure oxygen in the hospital, you aren’t. It is diluted with air every time you breathe in via the holes in the side of the mask. In most cases, as we mentioned in part two of this series, you probably aren’t even being given 100% at source either but more likely 60%. Your bed sheets are getting more oxygen than you are as it spills from your mask when you breathe out, and is diluted via the vents when you breathe in. Even if 100% oxygen is delivered to the mask it is almost always diluted considerably on inspiration. Only a tight fitting oral nasal mask with a demand valve can deliver a proper 100% oxygen. The kind we use for HBOT.

The number of allowable OTU’s for a 24-hour period varies with different analysis methods and is subject to several contributing factors.
These factors include things like, single exposure, multiple exposures, extended repetitive exposures, and compound residual build-up of the intolerance effect as well as of course, personal susceptibility factors.
Depending on which tracking method is subscribed to, a table of pre-determined exposure limits will likely be followed based on an upper limit on the number of OTU’s a person can tolerate in a single 24-hour period, or a table based on the cumulative effect of multiple exposures over many days. These tables are often tabulated into a single, dual purpose exposure limit table.
Somewhat beyond the scope of necessary information for patients, and more applicable to the provider of the HBOT therapy, to make proper use of the oxygen toxicity tracking tables one must first calculate the number of OTU’s in each exposure. This is done by considering the partial pressure of the oxygen being breathed, (ppO2), as discussed in previous articles, and the duration of the exposure. The following formula is the standard OTU calculation formula: (Tolerating Exposure to High Oxygen Levels – Repex and Other Methods Hamilton 1989)
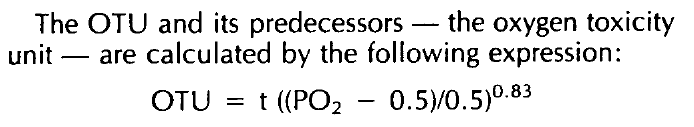

The above formula and calculation comes from our favoured tracking method, the Repex Method. The PDF is available on the page https://cumbria-hyperbaric.org/files-and-resources-download/

A recommended read for those interested in, or engaged in hyperbaric medicine, specifically medical professionals who haven’t encountered it before. Or even for those enthusiasts of science among us who have an interest in such things.
The Repex method follows a principal of whole body toxicity tracking as opposed to tracking CNS (cerebral) toxicity and pulmonary toxicity independently. It is a particularly useful method when tracking the cumulative effect of multiple exposures to lower pressures of oxygen over multiple days in diving or hyperbaric oxygen therapy. For the purposes of HBOT and lower level exposure over longer duration’s, and multiple days and cumulative effect, and volume loss due to lung absorption, the Repex method is favoured.
The lung damage mechanism most agreed upon is that prolonged exposure to raised partial pressure of oxygen degrades the surfactant found lining the walls of the alveolar sacks. Surfactant is the moist substance lining the lungs which facilitates oxygen transport through the alveolar wall. This is described by some as exposing lung tissue to excessive oxidation and in the diving industry some refer to this as “burning” the lungs. This is not an entirely accurate description of the mechanism responsible for causing lung damage. There is also usually mention of the formation of dangerous oxygen free radicals in lung tissue when breathing pure oxygen. Another cited cause for this intolerance is a condition known as lung absorption collapse. (Oxygen and The Brain – The Journey of Our Lifetime – James 2014)
In his book, “Oxygen and the Brain – The Journey of our Lifetime” – 2014. Professor Philip B James explains this extremely well. Far better than I ever could. Basically, he suggests, and we subscribe to this, that:
“Despite a lack of evidence, it is a commonly held belief that the effects of oxygen on the lungs are due to the formation of the ever-fashionable free radicals and that they are produced directly by breathing a higher level of oxygen”.
He goes on to add that this is believed despite good evidence in recent years showing this to be untrue. He adds that despite divers breathing increased oxygen well in excess of what would be 100% at sea level, they do not suffer lung absorption collapse either. Seemingly evidence of effectiveness is required in all aspects of trials, but not so in this case. Despite there being no evidence oxygen causes free radicals, it is still the default position. A double standard perhaps?
Lung absorption collapse is described as a lowering of lung volume due to metabolism of oxygen in the lower reaches of the lower lobes of the lungs. Whilst breathing pure oxygen, the inert gas portion of air, nitrogen, is flushed out leaving only oxygen to keep the lungs inflated, which is then metabolised lowering its volume. It absorbs directly through the alveolar wall. Ordinarily the nitrogen is not metabolised and remains in place as a sort of splint for the support of the alveolar sacks. This then causes those areas of the lung to partially collapse, attracting neutrophils and white blood cells to the area which themselves are designed to de-construct tissue resulting in tissue damage. This is what they are designed to do in fighting what they perceive as injury or infection, resulting from the now hypoxic state of the surrounding tissues following any such collapse.

This can only occur however after many hours of breathing pure oxygen at specific pressures. As we repeat a few times in various articles, we go nowhere near this level of exposure despite common perceptions. This is the other argument in favour of air breaks. By taking an air break, the lost inert nitrogen normally present in the lower reaches of the lungs is replaced. Without an air break, during oxygen breathing periods this nitrogen is displaced. As the body metabolises the now pure oxygen present, and the volume decreases in those alveoli, lung absorption collapse may be possible. This can then lead to localised hypoxia in the lung tissue. For more on hypoxia the article on hypoxia is an interesting read. HYPOXIA – THE COMMON DENOMINATOR IN DISEASE, INJURY AND ILL HEALTH
This causes a reaction in the now hypoxic tissue which signals the hypoxia response discussed in the article ALTITUDE, THE HYPEROXIC HYPOXIA PARADOX, AND THE COMMON SENSE PARADOX. This includes the immune response and tissue repair response as well as inflammatory response.
Remember that the body “thinks” there is an infection or injury because the tissues have become hypoxic following some measure of alveolar collapse. It’s this response that causes the damage. It’s at best, an indirect consequence of breathing pure oxygen and not a direct response, as is so often touted.
By replacing some of the nitrogen during an air break, the inert gas acts as a splint and keeps those areas of the lung inflated, preventing lung absorption collapse and any consequent misinterpreted immune response or hypoxia induced response.

Prof. James goes on to explain the mechanism of the air/blood barrier present in the alveoli and how it becomes compromised allowing plasma from the blood to leak into the lungs, eventually resulting in pulmonary oedema which can be serious indeed if untreated. By taking short air breaks this is avoided. Certainly, at the level and within the exposure time frames commonly found in HBOT.
He also establishes in his book that any damage that does occur, occurs because the air/blood barrier is compromised. This attracts neutrophils to the area, which stick to the capillary linings, and release free radicals into tissues on the blood side of the barrier as they are supposed to do. In effect this means that any damage that does occur, is not a result of direct poisoning of the lungs directly caused by oxygen breathing, but rather an immune response triggered on the blood side of the alveolar wall. Not direct toxicity at all but a physiological response to what is perceived as infection or damage owing to local hypoxia following alveolar collapse.
Returning to the Repex method cited, and now that we can calculate the number of OTU’s attributable to a single or repetitive exposure/s, as speculated above, we can now refer to the limits suggested by the Repex curve table and the Repex OTU tables reproduced below:
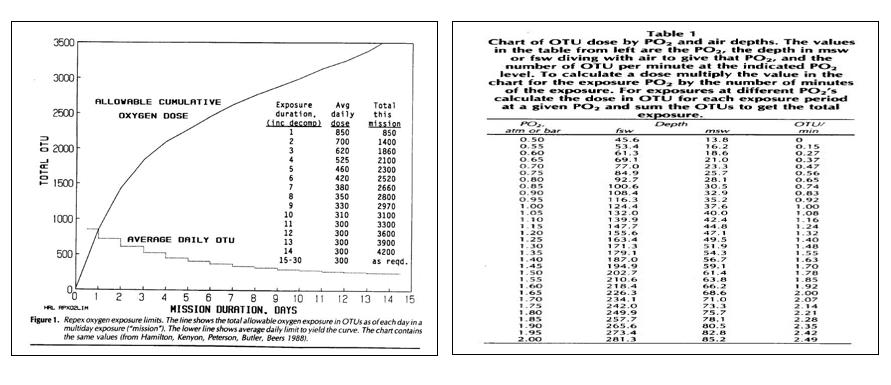
These two tables tabulate the limits suggested as the upper most limits.
The first table shows that over a longer period, the permissible daily OTU limit comes down from as much as 850 units for a single one-day treatment to around 300 OTU’s per day over a 15+ day period.
If memory serves, and as calculated using the Repex method, the standard US Navy table 6 for divers with decompression illness will incur an OTU penalty of about 600 units, and as such is within the tolerable limit for a single day. (Depending on extensions, which we do in diving therapeutic treatments), this table may exceed 600 OTU units but is still likely to remain within the tolerable 850 OTU units allowed). Accordingly, even a seven hour plus diving treatment remains within tolerable limits. Clearly any shorter HBOT session of 90 minutes, and lower pressure would be entirely within any conceivable limit.
Aside:
600 OTU units was considered the original safe single exposure limit imposed by many diving agencies. As mentioned above this is the OTU penalty incurred during a standard table 6 re-compression treatment. This table is incidentally extendable, or it can be converted to other tables such as the table 4, and the Comex CX30 treatment table. In cases of extensions or saturation treatments, intermittent oxygen breathing periods or lower concentrations of oxygen would be used to avoid toxicity response. The whole-body toxicity approach allows for an 850 unit upper limit as opposed to the original approximately 600 unit limit for pulmonary toxicity alone.
The second Repex limit table above is a table showing OTU’s per minute of duration of exposure. It saves one calculating for every depth. Once the pressure of oxygen is known, cross the table to learn the OTU per minute penalty, and with that multiplied by the time of the exposure and total OTU count for that treatment/exposure can be calculated. For exposures over many days, refer to the first table for the daily limit allowed for the predicted number of days or repetitive treatments and cumulative effects.
Inside these limits any dry cough, chest pain under the sternum, lung irritability etc will reverse readily following cessation of oxygen breathing.
Note: The lower limit, (and formula factor of 0,5ATA), is the accepted level of partial pressure of oxygen under which no significant damage will occur. This means that oxygen can be safely breathed indefinitely at a ppO2 of 0,5 ata and less.
This is incidentally the pressure that was used in the early days of space exploration with the Apollo spacecraft being pressurised to just 5 psi, about a third of atmospheric pressure. (NASA History Online) NASA routinely uses pure oxygen atmospheres, specifically in extra vehicular suits (spacesuits). To pressurise with air, one would need to pressurise beyond a pressure which would allow comfort and flexibility in the gloves. Hence lower pressures are used and accordingly oxygen concentration must be increased to maintain a suitable partial pressure of oxygen but still balanced with a pressure at or below the threshold of 0,5 ata oxygen partial pressure, under which no damage occurs to lungs breathing pure oxygen.

It is more common nowadays however, as the duration of missions becomes longer and longer than the early missions, with astronauts taking up residence in the ISS for months at a time, and as much as a year or more, for atmospheres inside space installations, to be either enriched to a lesser degree or to be comprised of air and pressurised to the standard 14,7 psi which equates to 1 ata. (The Space-Flight Environment: The International Space Station and Beyond Thirsk Et al 2009)
Hyperbaric operations, diving, and specifically saturation chamber technology and saturation diving and treatment physics and physiology, make use of the same physics as those used in space exploration. More can be read on this in the article SPACE TECH AND HYPERBARIC TECH: TECHNOLOGICAL COUSINS. The similarity between orbital habitat modules, or the proposed Mars or Lunar habitat modules, is uncanny. They even look like saturation complexes and hyperbaric chambers with the exception of being controlled by the occupants from the inside. Space shuttles, habitat modules, rovers, and even space suits are basically hyperbaric chambers using a pure oxygen or enriched air internal atmosphere. They are kept at lower than atmospheric pressure when using pure oxygen to avoid oxygen toxicity.
Accordingly, when considering the upper Repex OTU limits, breathing pure oxygen at sea level (1ata) cannot be safely done indefinitely. If actual pure oxygen is administered via a tight fitting oral nasal mask at normal atmospheric pressure, and a patient does indeed receive undiluted 100% oxygen, OTU limits must be observed. Th example below illustrates what time in a 24 hour period would be safe on proper pure oxygen.
As we can calculate:
Breathing pure oxygen at sea level for 1 minute as discussed, incurs 1 OTU. Since there are 1440 minutes in a day, a 24-hour period breathing un-diluted pure oxygen at sea level, would incur an OTU penalty of 1440 OTUs. Well in excess of the recommended 850-unit upper limit for a single exposure, and certainly above the former limit of around 600 units. This why many hospitals may tell you you are breathing oxygen when you are actually breathing medical nitrox. To continuously breathe mask supplied enriched medical air or nitrox for a full 24 hours, the maximum per minute allowable OTU would be 0,59 OTU’s per minute to not exceed an 850 unit per day penalty. Less, if multiple days exposure duration is predicted. According to the second Repex table this would mean the oxygen fraction in the breathing mix should not exceed 75% at sea level, an important thing to know if you or a loved one ends up on actual pure oxygen administered by hospital staff in training or by those not fluent in diving medicine. Notwithstanding that, as we discussed above, standard oxygen equipment doesn’t deliver 100% oxygen anyway.
An argument often made by medical professionals is that anything more than baric oxygen will become toxic. In the case of breathing high concentrations for 24 hours at a time, and indeed days at a time in a spacecraft, this would be true. In the case of HBOT though, we don’t routinely treat for more than 2 hours at a time and go nowhere near the times required for pulmonary toxicity to occur, rendering that argument defunct.
It’s worth noting that the most commonly used treatment tables for decompression illness (US Navy Rx tables 5 and 6) begin at 18 meters/60 feet (2.8 ata) breathing pure oxygen for 20 minutes. This is a recommended “go to” course of action taught in supervisor training and affords the supervisor the time to pull out tables, get his/her head in the right place, call for medical help and make appropriate decisions on how to proceed in an emergency. It is the depth at which the recommended upper limit of oxygen happens to be (2,8 ata), it is also the start point for many other therapeutic re-compression tables enabling an acceptable crossover to most other tables from that point if required, as well as extensions and cross over to saturation treatment tables. It is also well beyond the exposure an individual would likely experience in routine HBOT.
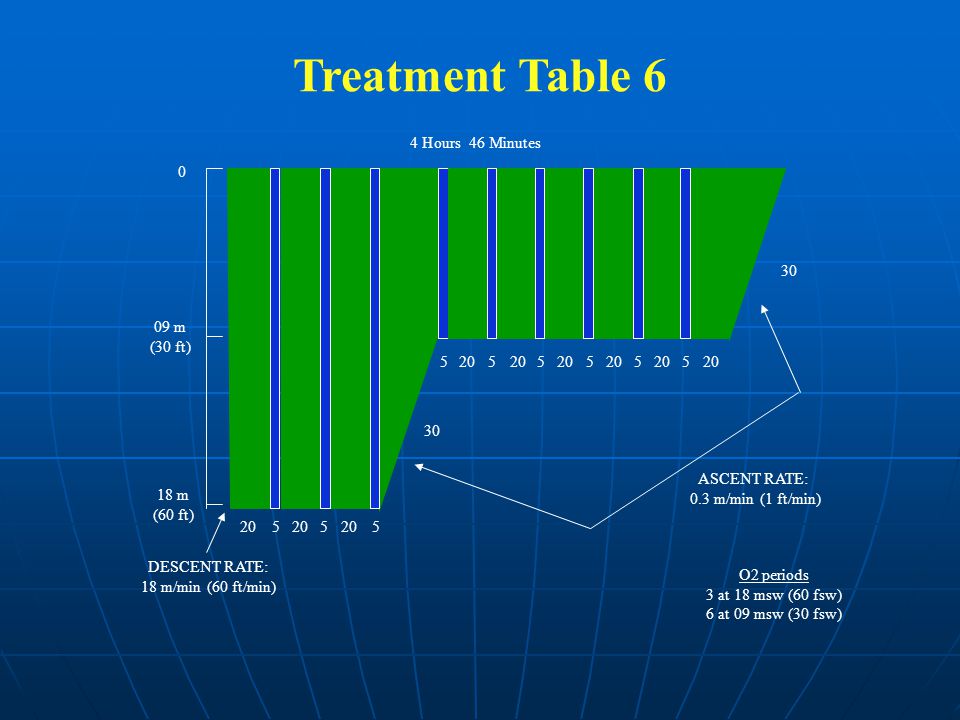
In fact, even the non-air table developed by Comex, the Cx30, which is a heliox mix of 50/50 (50% helium and 50% oxygen), treatment table can also be crossed to from this 18-meter start point. It only requires further compression to 30 meters and a gas switch. It is also the standard approach to DCI as defined by the US navy. Certainly, a good emergency starting point. It may seem less than relevant to discuss re-compression tables for decompression illness however it is simply the case that weather clinical compression for HBOT, or diver re-compression for decompression illness, both applications are considered to be hyperbaric oxygen therapy (HBOT).
From this information, we can calculate that even a relatively high treatment pressure of 3 ata for 75-minutes could be tolerated for 5 consecutive days with little risk of incidence. It follows then that any lesser protocol would be well within limits. Especially considering that treatments are often delivered over 5 days with 2 days off between 5 day runs.
A standard 2 ata / 90-minute protocol can be tolerated in excess of 15 consecutive days. Although that is rarely done with days off every 5 or 6 days. Any mild symptoms which present will likely subside when staying well below the curve.
Hopefully the above considerations, and those in the previous two installments demonstrate adequately the safety of hyperbaric oxygenation as it pertains to oxygen toxicity, or at least raises the question and possibility that what have been hard held beliefs don’t necessarily apply to these intermediate levels of supplemental and hyperbaric oxygen. No one is disputing that oxygen can and does have a toxic effect in extreme exposures, what is being stated though is that these exposure levels are generally never exceeded in standard HBOT and even in re-compression therapy for divers.
There is absolutely no reason to deny or discourage therapy based on the fear of toxicity. It simply isn’t the case.

©Hayden Dunstan


1 thought on “Oxygen Tolerance – Part Three – Pulmonary Oxygen Toxicity & The Repex Method”