
Building on yesterday’s sojourn into physics and physiology, it is important to elaborate on the third main gas law affecting divers, and by extension, and probably the most relevant of the laws, to hyperbaric chamber occupants. Yesterday we discussed briefly the effects of Dalton’s Law and Henry’s Law in the article entitled ‘Flying and Diving – A Sojourn Into Physics and Physiology’, to be found here:
https://cumbria-hyperbaric.org/2019/07/23/flying-and-diving-a-sojourn-into-physics-and-physiology/
Today it’s the turn of Physicist and Chemist Robert Boyle who published his law in 1662. Boyle’s Law.

The laws discussed yesterday related largely to their effect on inert gas as described by the two laws. They also lead into the mechanics of oxygen absorption in higher concentrations which is the underlying mechanical basis for HBOT and the hyper-oxygenation of blood and tissues.
Gas spaces in the body present specific points of concern in the hyperbaric environment. Explained by Boyle’s Law, pressure has a direct and inversely proportional effect on these air or gas spaces. This can lead to injuries caused by the reduction in the volume of a space, or indeed the increase in volume of a space. This is known as barotrauma. It can lead to serious problems for chamber occupants and divers alike if not managed properly, and can include inner and outer ear barotrauma, lung over-pressurisation injury, sinus squeeze, general squeeze and reverse block. This extends to air spaces contained within medical devices such as some cardiac pacemakers.
Barotrauma is defined as tissue damage in the body as a result of a differential pressure between the environment and the space in the body. The most common complaint being ear barotrauma or ear drum injury.
Every physician involved with hyperbarics should be familiar with the principals of pressure, manifestations and clinical management of pressure related problems.
Boyle’s Law States:
“For a fixed amount of an ideal gas kept at a fixed temperature, pressure and volume are inversely proportional.”
In layman’s term this means that as pressure increases, the gas in a flexible container such as the lungs or a balloon will decrease in volume. Conversely, when pressure is reduced the volume of a gas in a flexible container will increase.
For this reason, all chamber occupants are told to never hold their breath and breath normally all the time. Especially during pressurisation and depressurisation.
In addition to the lungs, other air spaces such as sinuses and specifically ear canals require equalisation during pressurisation or depressurisation. This adds or removes volume to or from the air space allowing the air to compress or expand with no consequence to the individual. Air compressing in the middle ear canal that cannot be equalised will decrease in volume drawing in the tympanic membrane (ear drum) causing discomfort and the potential to rupture to the ear drum. The same can happen in reverse when pressure is decreased causing expansion of the air inside the middle ear canal. If this expanding air becomes trapped it can lead to complications. This can be referred to as a block when pressurising, or a reverse block when depressurizing.
Similarly, sinuses which cannot be equalised can lead to pain, sinus “squeeze” and even some bleeding, sometimes presenting as a bloodied nose or slight bruising around the eye or indeed, a bloodshot eye. All in all, not overwhelmingly serious in nature.
In most cases this amounts to minor discomfort not dissimilar to going up in an aeroplane. The same fullness of the ears is common to both experiences.

Equalisation of these air spaces is imperative. Whats more though, it is not difficult at all and anyone can do it. The ears can be equalised by executing the Valsalva manoeuvre, (blowing gently against blocked nostrils or airway).

This manoeuvre forces air up the Eustachian tube inside the ear and equalises the volume of the air space. When we say equalises, we mean that it equalises the volume and pressure between pressure outside the ear and pressure inside the ear relieving any expansion related discomfort. Wiggling the jaw side to side, or extending the jaw forward slightly also works well as it does in equalising during flying.
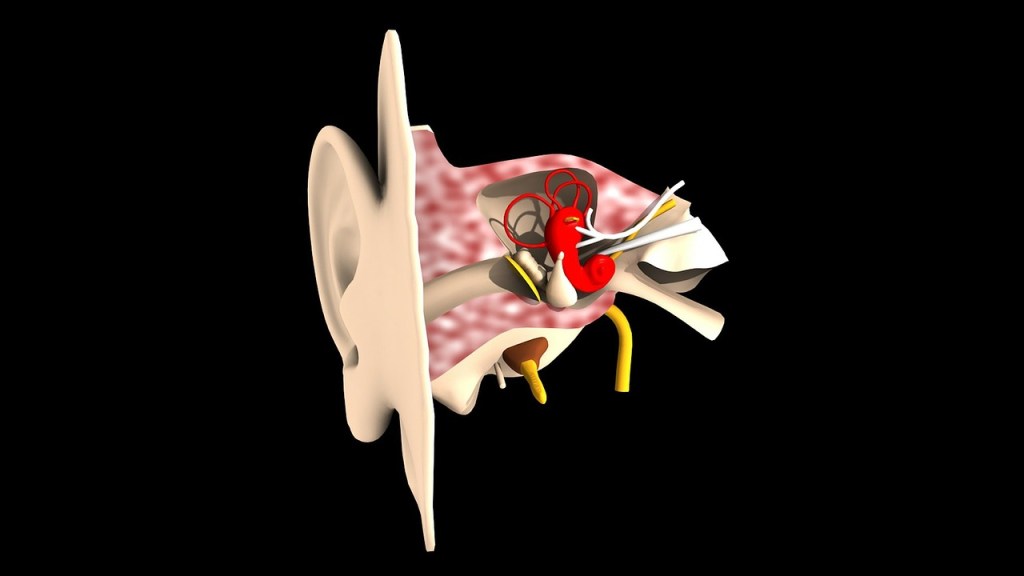
Air spaces in teeth are harder to equalise and very rarely divers experience dental discomfort or barotrauma. Barotrauma simply means trauma or injury caused by barometrical (pressure) changes. Dental health is always highly recommended in divers for this reason. It is extremely rare for chamber occupants to have any serious issues. In any case, if someone does experience a problem we will stop the pressurisation immediately until we can resolve the issue. The worst that may happen is a filling may come lose. Unfilled teeth present no problem as there is no air space present.
Gastro-intestinal gasses simply compress and re-expand with no consequence. Perhaps with only one exception. It is not uncommon to pass wind or belch upon de-pressurisation as gas expands. It is very common to swallow air during a treatment or a dive, however, this will cause no real discomfort and is of little consequence as it will resolve itself.
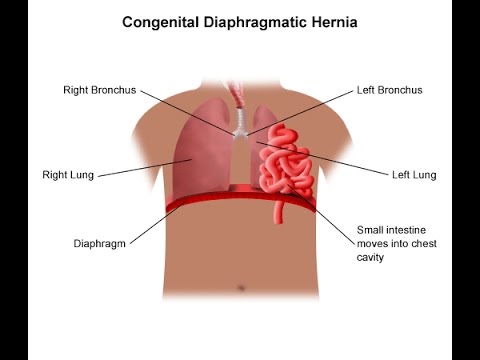
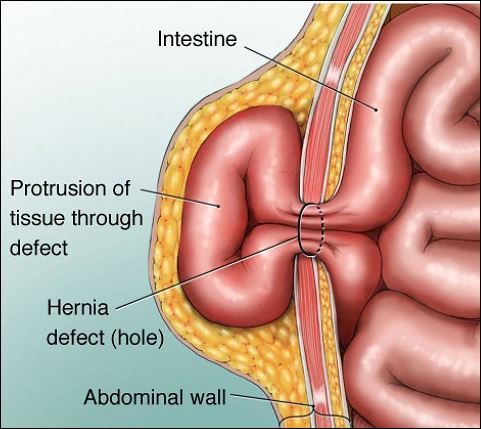
The one exception to the above is un-repaired hernia. Individuals with un-repaired abdominal wall or diaphragmatic hernia are not suitable candidates for pressurisation until after it has been successfully been repaired. rare as it may be, swallowed air may become trapped in an intestinal protrusion and become trapped, presenting potentially dangerous consequences when that air begins to expand as pressure is reduced.
The above stated physical law was confirmed by Robert William Boyle (25 January 1627 – 31 December 1691), an Anglo-Irish physicist and chemist. He confirmed the earlier work on the relationship between pressure and volume which was first noted by Richard Towneley and Henry Power in the seventeenth century.
Ruling out any temperature changes, this law defines the relationship between pressure and gas volume.
Diving instructors across all disciplines will drill this into any candidate undertaking a diving course, or any other course such as, sub-marine escape training, tunnelling (under pressure), caisson worker, air lock operator etc. Many of these disciplines are unrelated to diving but fall into the mining disciplines.
A word on tunnelling: Tunnelling works are often done in a compressed air environment to keep ground water from seeping into the works area. This is essentially the same as breathing compressed air whilst diving, or indeed in a hyperbaric chamber. Air is breathed under conditions exhibiting the same volume / pressure relationship. In a flexible container, volume is subject to Boyle’s Law. Volume behaves inversely proportional to pressure. In fact, many tunnelling workers working in a compressed air environment are decompressed just like divers at the end of their shift or period of exposure.
The primary consideration of this law is that a flexible container will expand along with the expanding compressed gas it contains as pressure is reduced. Conversely, the gas a flexible container contains, will be compressed along with the container as pressure increases, i.e. a balloon filled under pressure, or pair of lungs or other flexible container, will eventually over expand if internal increasing volume expansion is not released or equalised when external pressure is reduced. This is classified as one of this risks associated with HBOT. It is however, very easily managed.
It’s common to take a few balloons into the chamber to demonstrate this relationship along with empty soda bottles. On deeper excursions it’s great fun to conduct experiments with table tennis balls as well.
An interesting and amusing side effect of breathing denser air is a rise in vocal tone. Sound travels differently through denser air, it travels faster making the voice pitch up a bit. Chipmunk voice can be amusing if the pressure is high enough. Certainly the case in 50 meter dry dives.
While lung over-expansion is a technical possibility, it is easily managed by breathing normally and not holding your breath. That simple. Patients with obstructive lung disease are subject to medical scrutiny as to suitability for HBOT and are generally contra-indicated if they cannot equalise the alveoli in the lungs. this forms part of the pre-assessment and informed consent. If required, we will ask you to visit your doctor prior to undergoing pressurisation.
Granted, we do have other air spaces in our bodies that are affected such as sinus cavities, middle ear canals behind the ear drum, lungs of course, and any such minor gas pockets as may be present in the digestive tract. Breathing normally protects the lungs, and with simple instruction, ears are easily equalised and sinuses take care of themselves. Gastro intestinal gas will simply compress and re-expand if a small amount of air is swallowed during a dive or treatment. This will assuredly find its way out along one of two paths with no ill effect, albeit a little embarrassing.
It’s worth noting that someone with obstructed airways who is unable to equalise may be contra-indicated until such time as their air passage ways are clear enough to equalise pressure changes. This includes cold and flu, congestion, hay-fever or other forms of congestion.
A risk to benefit assessment should be done on patients requiring emergency treatment where avoiding or delaying treatment may result in a worsening of their condition. Sometimes the sacrifice of an ear drum is prudent. Especially in cases of decompression illness and arterial gas embolism.
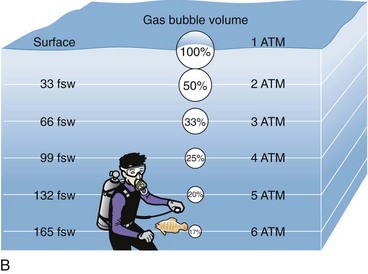
Pressure experienced by increasing water depth is the same as pressure experienced in a hyperbaric chamber. It’s the reason we often refer to chamber sessions as ‘dives’ and chamber pressures as ‘depths’. Chamber sessions are often referred to as ‘dry dives’ and the pressure simulates depth under water. 1 atmosphere (ata) of pressure is the equivalent of 10 meters of sea water as shown in the diagram.
Since the remainder of our body is largely water and generally of a similar density to water, pressure has little or no effect on it in depths shallower than 700 meters or 71 ata. The body itself outside of air spaces is largely incomprehensible and is sensationally no different from normal pressures.
More information is available in the overview on physics and physiology.
Always remember, never hold your breath.
Hayden Dunstan



Hey,Good post. I learn something totally new and challenging on sites I stumbleupon everyday. thanks
LikeLike