
Borrowing from previous articles reproduced here is a definition of Hyperbaric Oxygen Therapy.
HyperBaric Oxygen Therapy is described as the medical administration of pure or near pure oxygen at higher than atmospheric (Normobaric) pressure.
As discussed in those articles, and explained previously, hyper-oxygenation is facilitated by an increased pressure differential or gradient between the partial pressure of oxygen in the breathing mix (gas phase), and the tension found in the tissues (liquid phase). As discussed in the article FLYING AND DIVING – A SOJOURN INTO PHYSICS AND PHYSIOLOGY.
When an increased fraction, (proportionate percentage) of oxygen is breathed under increased pressure, it will dissolve in greater amounts into the blood according to Henry’s Law. (Pulmonary Gas Exchange – West Et al – 1998).
This is not to say that the haemoglobin will suddenly be able to carry more oxygen, not at all. It means that the blood plasma becomes saturated to a higher degree. This results in a far greater saturation of oxygen in blood plasma and in turn results in greater saturation of individual tissues and cells in the body. In mechanical terms, this is achieved by increasing the ambient pressure surrounding a persons body, facilitating the breathing of oxygen at increased pressure while maintaining pressure equilibrium inside and outside of the body so as to avoid disbaric injury or pressure differential injury. By doing this the individual is almost entirely unaware of the change in pressure, save for the effects on air spaces such as the ears.
This can usually only be achieved by making use of a sealed chamber capable of being pressurised and depressurised at will. That is, a hyperbaric chamber also called a pressure chamber. Since the body, other than its air spaces, is predominantly water, it is for all intents and purposes in-compressible and this pressure change is not noticeable. Even in very deep experimental dives by Comex in France, to 701 meters in saturation chambers, the occupants’ bodies tolerate the pressure well. (Diving Almanac Hydra 10 2018) (Deep Hydrogen Diving Hydra 10 Gallant 1993)
This effect can also be achieved by diving under the water. Hence the alternative name for a hyperbaric chamber, namely, a diving chamber. Diving physics explains that for every 10 meters of sea water a diver dives under, an additional one atmosphere of pressure is added to their ambient pressure, i.e. 10 meters of sea water (MSW) is equivalent of 1 ATA. This is much the same as the weight of the atmosphere previously discussed in the above article on Flying. Water however, being far denser, about 800 times denser than air, exerts a far greater weight or pressure than air. Therefore, as little as 10 meters of water is equal to full 10 000 meters of the measurable pressure of the atmosphere in terms of weight per unit of similar area. Not to forget the actual atmosphere above the water, which is also equal to one atmosphere. This adds to the water pressure to give a measurement of absolute pressure. (ATA, or atmospheres absolute).
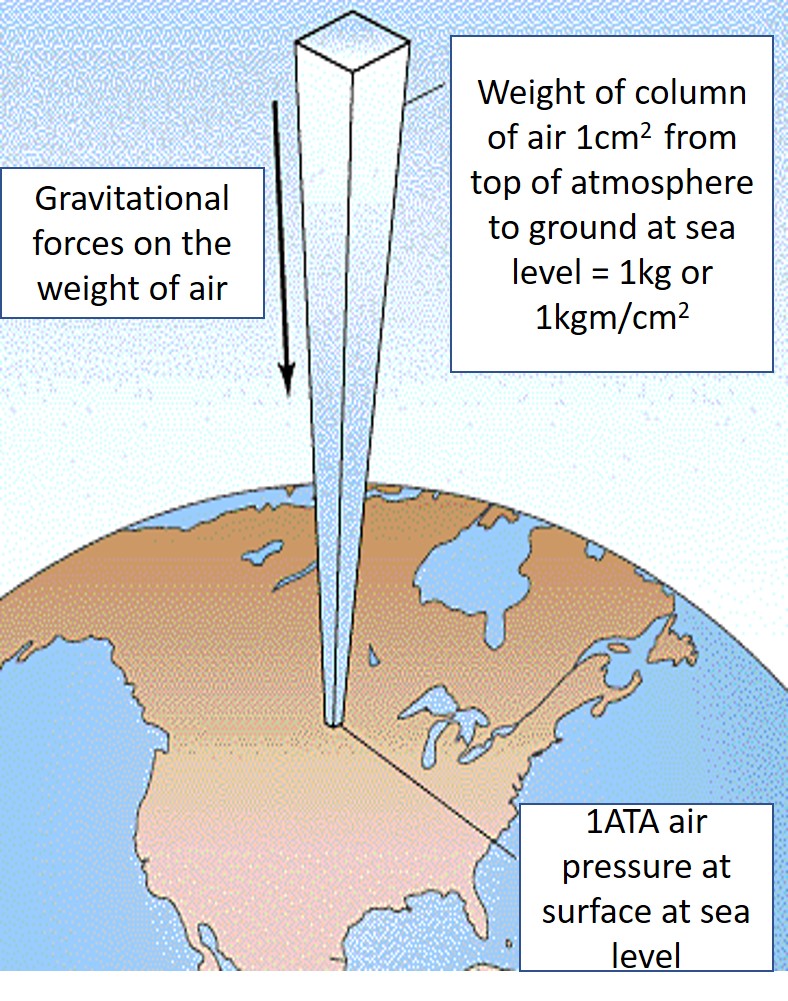
Accordingly, at a depth of 10 meters, a diver would be under 2 atmospheres of pressure, (1 for the water pressure and 1 for the actual atmosphere), at 20 MSW, 3 ATA and so on. 2 ATA being a common pressure used in HBOT protocols and therapy plans. These calculations are only really important for operators, divers and supervisors to understand, as they pertain to gas flow and volumes as well as inert gas absorption and other gas management factors, but not so important to understand as a patient. Doctors also need to have an appreciation for the ambient pressure at which oxygen is being breathed. It is an important factor in determining protocols and treatment dosages for each patient. Some conditions require higher pressures than others. Some require longer treatment regimens as well. More details can be read in the article DOSAGE: HBOT AND MHBOT.
The technology originates in the diving sector and has gradually crossed into the medical world and shown itself useful in a variety of medical applications. Many divers and supervisors noticed a curious correlation between medical physics and diving physics. They noted that some of the things we are warned about, and taught, could serve as therapy for some medical conditions, if not as a cure then certainly as a management tool, and complementary therapy to control symptoms and provide better quality of life. Extensive study since those early days have indeed shown that diving chambers can benefit many medical conditions outside of the traditional “Bubble Squashing” use in diving.
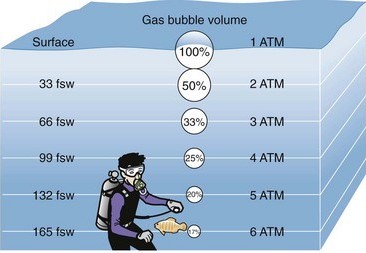
By ‘bubble squashing’, I mean the actual squashing of bubbles which can be present in cases of decompression illness as described above. Henry’s Law determines the solubility of a gas in a liquid under pressure. In the case of metabolically inert gas, it will simply come back out of solution when pressure is reduced, as in, when a diver surfaces. This can result in the onset of decompression illness. Essentially it is what would have been called; the bends, decompression sickness, caisson disease and so on. Boyle’s Law (diagram above) dictates that increasing pressure in a chamber will reduce the volume of any bubble present and quite literally, the bubble is squashed.
This is not the case with oxygen. Oxygen cannot embolise as it cannot form a bubble meniscus in the early stages of bubble formation following the development of the micro-nuclei. Only inert gasses come back out of solution to any significant degree in this manner. Another reason for this is that oxygen is metabolically active and is metabolised (gets used up), to an increased degree when breathed at higher than normal pressures. Explained in other words, as the pressure of oxygen increases so does the oxygen tension in tissues and cells. (Tension is simply the word we use for ‘pressure’ when its in solution in blood and tissue). With this, there is an increase in the metabolic process which consumes the oxygen. The glucose cycle is up regulated as is the entire metabolic process. Conversely, inert gas does not get used up and remains in solution in its original quantity until the pressure is relieved. Much like the gas in fizzy drinks. Since we largely don’t breathe any inert gas during a therapeutic session, inert gas is of no consequence.
When a diver suffers from decompression illness, the increased pressure of a re-compression treatment simply squashes the bubble/bubbles according to Boyle’s Law and re-dissolves the gas into solution in the tissue or blood according to Henry’s Law. This is followed by a gradual reduction in pressure to avoid a re-occurrence of the aforementioned micro-nuclei and bubble formation. It is facilitated by breathing pure oxygen in pre-determined intervals or periods know as treatment, Rx tables, or treatment protocols as they’re know to HBOT operators.
This is Hyperbaric Oxygen Therapy in its simplest form and is the only indicated treatment worldwide for decompression illness.
Both the U.S. and Royal Navies have developed a wide range of Rx tables after more than half a century of research, (and before the days of stricter ethical controls), experimental dives with actual human subjects. The best analogue so to speak. They also collect and analyse data from millions of dives acquired from voluntary data sharing from divers around the world. These tables and data are universally accepted and extensively used today in commercial, military and sport diving. (United States Navy Diving Manual – Underwater Physiology and Diving Disorders 2016). In fact all diving tables available today come from the same source. The original Buhlmann calculations which are used in military, commercial, technical and recreational diving around the world. The only variation really amounting to different levels of conservatism. Find out more about Albert Buhlmann on the link.
https://en.wikipedia.org/wiki/Albert_A._B%C3%BChlmann
This technology has shown to not only to help decompression illness, but a host of other afflictions as listed on the page entitled “Indications For HBOT” https://cumbria-hyperbaric.org/indications-and-conditions-we-plan-to-treat/.
It’s a simple concept. The primary requirement for life is oxygen. Without it life ceases fairly quickly. The primary requirement for biological process is oxygen. Without it, cell function ceases and the natural growth, repair and rebuilding processes in the body cease. Giving more of it increases cellular metabolism and enhances the body’s natural process of healing and repair. The body is its own pharmacy. It has evolved to maintain itself. In cases where normal oxygenation isn’t quite enough, hyperbaric oxygenation (hyper-oxygenation) can be administered to simply enhance what exists in nature already.
After taking the long way round to describing the hyperbaric environment, chambers themselves are usually made of steel and are great big tubes with dished or flat ends, with small viewing portholes much like a submarine from a children’s story book. They are intimidating to the inexperienced to say the least. Others are made of transparent acrylic and are essentially one-man tubes in which a patient can be observed from the outside. In fact, I’m told these acrylic chambers were developed to allow for radiotherapy to continue while conducting HBOT treatments to better protect the body from the negative effects of radiation.

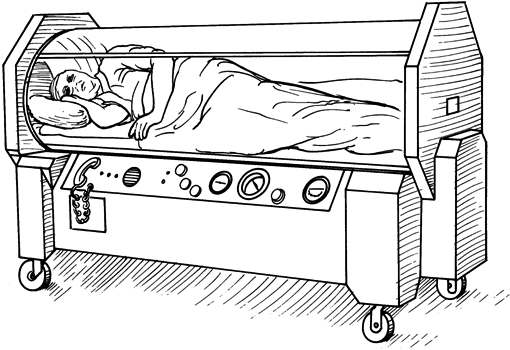
The big chambers for more than one person are referred to as multi-place chambers and the one-man acrylic ones are referred to as mono-place chambers for obvious reasons. In diving and diving related emergency treatments, we use what are called twin lock chambers. This simply means it has two compartments. The entry lock (compartment) and the main lock. They have the same set of controls for each compartment inside and outside and they can be equalised with each other. These are called double skinned valved chambers. It allows an attendant to take control of the treatment from the inside if needed. The entry lock serves as a method of personnel entering and exiting the chamber without the need for decompressing the chamber also called surfacing the chamber. Much the same way an astronaut enters and exits a space shuttle or the space station. It also allows for the delivery of medicines, food, blankets and other materials as may be needed during a treatment without surfacing the chamber. They are usually fitted with a third smaller compartment called a medical or ‘med’ lock or transfer lock, which can be used to transfer smaller items in and out of the chamber without surfacing the chamber. This allows for uninterrupted treatments.
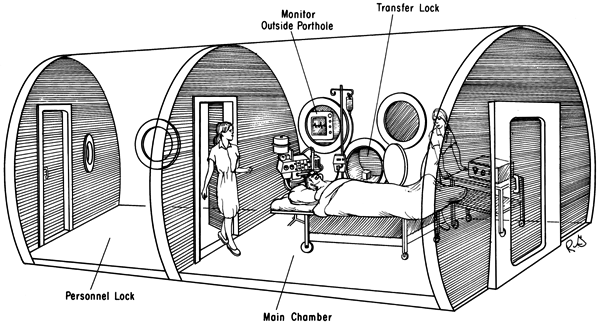
In medicine, chambers differ a little, but the principal is the same. Some facilities will use mono-place chambers as described above and some will use multi-place chambers as described but with only one compartment. They are usually used for what is termed “ambulant” patients who are not in a condition requiring emergency handling and treatment. These single lock (compartment) chambers are more affordable and easier to run than their more advanced cousins. They are ultimately best suited to “walking, talking, voluntary patients”.
UK law describes these as type 3 chambers, and in 2008 they were deemed so safe that they were deregulated by an act of parliament under the amendment of that year to the Private and Voluntary Health Care (England) Regulations 2001. Although some risk is present, in the millions of treatments conducted by non-medical staff and volunteers in the last 30 years, there has never been a reported serious incident.
Hospitals have chambers which are a combination of both of the above. They have what are termed by the UK Health Department and the Cox Report as type 1 and 2 chambers, and those chambers can offer emergency referral diver treatment as well as type 3 HBOT, as opposed to a type 3 chamber which may only provide elective complementary therapy type 3 HBOT under the type 3 amendment.
Currently the law allows for type 3 HBOT services to operate without Care Quality Commission registration, for which these types of services do not qualify. The authorities recognise them as complementary therapy rather than purely medical treatments.
Facilities which have two compartment chambers are capable of offering, in the case of type 1, advanced life support capability, and in the case of type 2, emergency life support capability. Type 1 chambers are required to be within a hospital facility that has helicopter access to offer a type 1 service, and type 2 is generally in a hospital, or sited near a hospital, that does not necessarily have helicopter reception facilities. A type 1 chamber must be sited within the hospital precinct. (NHS Commissioning of HBOT 2013)
In keeping with the main heading of this article, the environment inside a chamber is actually rather fun and cool. Contrary to misconceptions, it is not particularly claustrophobic or cramped, even when holding lots of patients. They are comfortable, safe, warm and often the type 3 chambers will offer TV or video facilities and so on. You can read, snooze or interact with your companions though your oral nasal mask. We discourage too much chatter as this tends to displace masks, meaning you may end up breathing air and defeating the intention.

In fact, it can be argued that one is safer in a chamber than out. When the inherent risk is managed properly nothing can hurt you inside a chamber. You are independent of the world outside. Much like David Bowie’s Major Tom sitting in his tin can. Ground control being your chamber operator on the outside. The old communication standard in the UK would have been something like “inside to outside… do you copy?”. To which the reply would have been “outside to inside… copy you five out of five”. Or something along those lines. These days it’s a lot less formal.
For those who have never been in a chamber, it usually starts with a briefing by either the nurse or attendant or even the operators themselves. They will instruct you on everything you need to know about safety, evacuation, what to do, what not to do, and so on. You will be monitored every second of your treatment. You will also be given a list of items prohibited in the chamber. This can be found in the article FIRE SAFETY AND PROHIBITED ITEMS – A NECESSARY DISCUSSION
Then comes what is called “blowdown” in diving. Divers have a knack for colorful descriptive language. This is merely the pressurisation of the chamber. The operator will talk to you via a communications system and keep you informed as to what to expect. If your operator is a diver like me, the communication may take a more formal tone, but with specifically trained HBOT medical type staff it can be a lot more relaxed. At first the operator will achieve a door seal and then stop to check everyone is OK inside, communicated either verbally or by use of hand signals. In diving we have occupants hold a thumb against one of the view-ports all the way through blowdown. That way it will be immediately obvious if someone removes a thumb indicating a problem. It can be a bit noisy, and on your first run maybe a little overwhelming, but it really is just sound of additional air entering the chamber. And yes, other than some small, low pressure acrylic mono-place chambers, we pressurise the chamber with air.
If your operator wants to pressurise a large multi-place chamber with pure oxygen, feel free to head in the other direction. Pure oxygen pressurisation of anything other than low pressure mono-place acrylic chambers designed for it, is not safe. (BHA Fire Safety 1996 & ECHM – A European Guide to Good Practice for Hyperbaric Oxygen Therapy)
During the blowdown the chamber will get warm. When a gas is compressed it warms up according to Gay-Lussac’s law. For the science enthusiasts among us this is as a result of adiabatic process. Next time you fill your car tyres at the garage, feel the tyre wall. It will have warmed up a little if they needed more than just a small amount of air, and if the weather isn’t too cold.
In saturation diving this is a serious consideration. SAT divers go to greater depths exceeding 100 meters and sometimes as deep as 300 or more meters compared to our 10 or 15 meters equivalent. If they blow down too quickly the heat can become problematic. In HBOT however we don’t go anywhere near these pressures and the warming of the chamber will be minimal but still noticeable. We also take it slow to afford chamber occupants ample time to equalise air spaces as we discussed under Boyle’s Law. The humidity may rise also. During the blowdown, any call to “STOP” will stop the pressurisation. If an occupant for some reason cannot equalise their air spaces, we will stop and reduce pressure until they can, then try again. While disbaric injuries are possible such as ear drum damage, they are extremely rare, especially with well trained staff managing your treatment and comprehensive ‘pre-dive’ briefings and induction for first timers.
The temperature will stabilise after reaching the treatment pressure or depth as it is so called. Since we simulate water pressure in a dry chamber, depth is the term favoured by divers. (Non divers may not be familiar with this term and refer to treatment or therapy pressure). The chamber will cool, and this will result in a slight lowering of the pressure in accordance with Gay-Lussac’s law above. This necessitates “topping off” the pressure to fine tune the treatment depth. This is known as “chamber creep”. It happens to dive cylinders too after they have been filled. They cool a little and the pressure drops requiring a “top off”.
You will then be asked, via the communications system, to don your oral nasal mask through which you will breathe oxygen. The oxygen will be delivered to you at the same pressure as the ambient pressure in the chamber. This is logical, as if it were less, it wouldn’t be able to pass into the chamber.
This is sometimes an area of confusion. No disproportionate pressure is applied to the lungs in any way. The overall ambient pressure is higher, and by virtue of this the gas being breathed is also at a higher pressure relative to the existing pressure or tension present in the body’s tissues, which is still equal to the pressure that exists outside the chamber. There is no differential present when breathing in a chamber. The pressure in the lungs is equal to the pressure in the chamber. It’s just like breathing in an aeroplane, which too is pressurised to a different pressure than what we are accustomed to.
Oxygen will come to you via a demand valve in the oral nasal mask, and it will deliver oxygen only when you breathe in, in the same way a diving regulator delivers gas to a diver. The action of breathing inward activates a diaphragm valve releasing the oxygen on demand.
When you breathe out, the high concentration of oxygen in your exhaled breath does not go into the chamber. It will pass through a second hose attached to the oral nasal mask and be vented outside the chamber. We call this overboard dump. It helps maintain a safe level of oxygen in the chamber atmosphere. As mentioned in other articles, chamber atmosphere should not be pure oxygen. In fact, it should not be allowed to rise above 23 or 24%. If it ever does exceed 24% your operator will likely prepare you in advance for a chamber “flush” in which they will vent the chamber and pressurise it at the same time. This will keep the pressure constant while replacing the chamber internal atmosphere with normal air thus maintaining a safe oxygen level. In diving we routinely flush chambers not fitted with carbon dioxide scrubbers or filters to keep carbon dioxide levels at a safe level. This has the dual benefit of ensuring safe oxygen levels also.
During your treatment we may give you what are called air breaks. This is a 5-minute period in which you will remove your mask and breathe the chamber atmosphere (air). If it is done, it would be every 20 minutes usually. We do it for divers also. We always have. Even those on extreme re-compression therapy tables.

Once your treatment time has elapsed we will inform you via the communications system that we are to begin depressurising the chamber or surfacing the chamber as it’s called. You may be asked to remove your oral nasal mask and close any associated valves. The attendant can help you with this. It’s simply a reversal of the blow down procedure. Air is vented from the chamber in a controlled manner, until it is equal with normal atmospheric pressure and the door can be opened. It can take a few minutes to do and sometimes a little longer for comforts sake. During de-pressurisation, any air spaces that now contain compressed air or other gas will expand and is vitally important to breathe normally and never hold your breath. That said, this is of little concern since we depressurise the chamber so slowly that almost no individual could hold their breathe long enough for it to become a problem in safe equalisation of the lungs.
Other air spaces such as sinus cavities and ears will equalise naturally on their own. In the event they do not, an occupant can experience what’s called a “reverse block”. The ascent will be halted allowing a little time to relieve the problem and then try again until we are able to reach the surface comfortably and safely. Bearing in mind that any individual has been breathing oxygen, and as such the oxygen in these air spaces has a far faster rate of absorption into tissue than inert gas, and it is consumed rather than than simply existing in solution. The ascent is slow enough for this to absorb entirely before becoming a serious problem. Any major signs of lung, airway or chest congestion or airway obstruction on the day will almost certainly exclude you from that treatment though. Basically, if we don’t think we can bring you out safely we won’t let you in. We’ll show you how the control panel works instead, and you can talk to all the Major Tom’s inside from ground control.
Following this, you go on your merry way until the next treatment. We ask you not to smoke for 30 minutes after a treatment. Indeed, as it applies to smoking anywhere near a chamber that is venting pure oxygen via the overboard dump. Your lungs will be full of oxygen and it takes a while to replace all that O2 with normal air again. Your clothes may also be a little “enriched” from the experience. They too “off-gas” in the same way people do. Porous fabric absorbs any oxygen it may come in contact with from incidental spillage from masks, give the fabric time to “off-gas” before lighting up. As you know, oxygen supports combustion. Many units may give you pocket-less scrubs to wear in the chamber for this purpose, among other reasons.
You will then probably enjoy the best night’s sleep ever. Explained in a little more detail in SLEEP – THE ELIXIR OF LIFE.
And that’s about it, although we took the long way around. It’s great fun and builds comradery among the folk that make use of chamber therapy.
©Hayden Dunstan

